SPECTROPHOTOMETRIC DETERMINATION OF PARACETAMOL IN PHARMACEUTICALS BY DIAZOTIZATION AND COUPLING REACTION WITH RESORCINOL AND 1-NAPHTHOL
Abstract
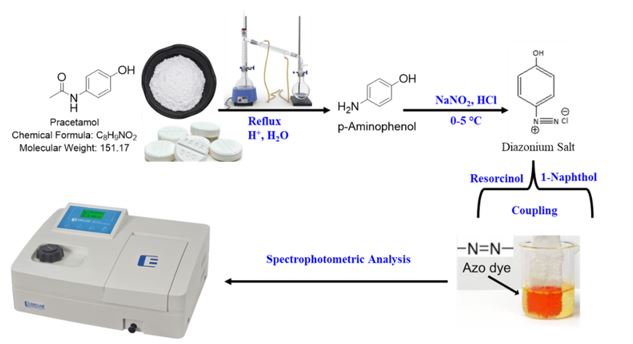
A simple, accurate, and sensitive spectrophotometric method has been developed for the quantitative determination of acetaminophen (paracetamol, PAR) in pharmaceutical formulations including tablets, syrups, and injectable solutions. The method involves acid hydrolysis of PAR to yield p-aminophenol, which is subsequently subjected to diazotization to form a diazonium salt. This diazonium intermediate reacts with resorcinol and 1-naphthol to form colored azo dyes, exhibiting maximum absorbance at 480 nm and 510 nm, respectively. The method adheres to Beer’s law over concentration ranges of 3–15 μg/mL for the resorcinol system and 2.5–20 μg/mL for the 1-naphthol system. The calculated molar absorptivity values were 18.5 × 10³ and 9.8 × 10³ L·mol⁻¹·cm⁻¹, with corresponding Sandell’s sensitivities of 0.008 and 0.015 μg·cm⁻², respectively. Method validation demonstrated satisfactory accuracy, with mean percentage recoveries ranging from 94.78% to 105.73%. Furthermore, the method exhibited good linearity, low limits of detection and quantification, and minimal relative error, underscoring its reliability and suitability for routine quality control analysis of PAR in commercial pharmaceutical preparations.
Full text article
References
Ahmad, N. R., & Omar, F. K. (2018). HPLC method for determination of paracetamol in pharmaceutical formulations and environmental water samples. World J. Pharma. Res, 7(15), 1-10. DOI:10.20959/wjpr201815-12814
Ahmed, R. K., Muhammad, S. S., & Khodaer, E. A. (2015). Spectrophotometric Determination of Paracetamol in bulk and Pharmaceutical Preparations. Baghdad Science Journal, 12(2), 317-323. DOI: https://doi.org/10.21123/bsj.2015.12.2.317-323
Arerusuoghene, A. C., & Sinodukoo, O. (2022). Solubility and dissolution optimization of paracetamol using in situ micronization by solvent change method. International Journal of Biosciences, 21(3), 8-19. DOI: http://dx.doi.org/10.12692/ijb/21.3.8-19
Arief S, M., Priya D, S., & Gayatri, S. (2023). Simultaneous determination of paracetamol and caffeine by RP-HPLC in soft gelatin capsules. International Journal of Science and Research Archive, 9(2), 950-957. DOI:10.30574/ijsra.2023.9.2.0691
AzEEz, K. F., Tahir, T. F., & Kokhasmail, D. M. (2021). New Reagent for Coupling Reaction and Spectrophotometric Determination of Paracetamol in Pharmaceutics. Oriental Journal of Chemistry, 37(4), 885-890. DOI : http://dx.doi.org/10.13005/ojc/370416
Bober-Majnusz, K., & Pyka-Pająk, A. (2024). Comparison of the Limit of Detection of Paracetamol, Propyphenazone, and Caffeine Analyzed Using Thin-Layer Chromatography and High-Performance Thin-Layer Chromatography. Processes, 12(6), 1153. DOI: https://doi.org/10.3390/pr12061153
Dewani, A. P., & Patra, S. (2015). A single HPLC-DAD method for simultaneous analysis of paracetamol, phenylephrine, caffeine and levocetirizine in bulk powder and tablet formulation: Application to in-vitro dissolution studies. Journal of the Chilean Chemical Society, 60(4), 2734-2739. DOI:10.4067/S0717-97072015000400019
Divya Vilas, K., Sandeep Reddy, C., Prashanthi, R., & Ankit, C. (2020). A Rapid LC-MS/MS method for quantification of acetaminophen in human whole blood. J Chromatogr Sep Tech, 11(5), 1-6. DOI: 10.35248/2157-7064.20.11.432.
Dixit, R., & Patel, J. (2014). Spectrophotometric determination of paracetamol drug using 8-hydroxyquinoline. International Journal of Pharmaceutical Sciences and Research, 5(6), 2393-2397. DOI: http://dx.doi.org/10.13040/IJPSR.0975-8232.5(6).2393-97
Ghazal, L., Almardini, M. A., & Allous, I. (2023). A VALIDATED RP-HPLC METHOD FOR SEPARATION AND DETERMINATION OF ETORICOXIB AND PARACETAMOL IN BULK AND PHARMACEUTICAL DOSAGE FORM. Bulletin of Pharmaceutical Sciences Assiut University, 46(2), 969-982. DOI: 10.21608/bfsa.2023.327630
Kam, R. K.-T., Chan, M. H.-M., Wong, H.-T., Ghose, A., Dondorp, A. M., Plewes, K., & Tarning, J. (2018). Quantitation of paracetamol by liquid chromatography–mass spectrometry in human plasma in support of clinical trial. Future Science OA, 4(8), FSO331. DOI: 10.4155/fsoa-2018-0039
Luu, N., Duy, T., & Binh, T. (2023). Simultaneous determination of paracetamol and codeine phosphate in pharmaceuticals using molecular absorption spectroscopy and classical least squares method. MOJ App Bio Biomech, 7(2), 41-46. DOI: 10.15406/mojabb.2023.07.00174
Mészáros, P., Kovács, S., Kulcsár, G., Páskuj, M., & Almási, A. (2022). Investigation of intestinal absorption and excretion of paracetamol in streptozotocin-induced hyperglycemia. International Journal of Molecular Sciences, 23(19), 11913. DOI: https://doi.org/10.3390/ijms231911913
Mezaal, E. N., Sadiq, K. A., Jabbar, M. M., Al-Noor, T. H., Azooz, E. A., & Al-Mulla, E. A. J. (2024). Green methods for determination of paracetamol in drug samples: A comparative study. Green Analytical Chemistry, 10, 1-13. DOI: https://doi.org/10.1016/j.greeac.2024.100123
Palakollu, V. N., Chiwunze, T. E., Liu, C., & Karpoormath, R. (2020). Electrochemical sensitive determination of acetaminophen in pharmaceutical formulations at iron oxide/graphene composite modified electrode. Arabian Journal of Chemistry, 13(2), 4350-4357. DOI: https://doi.org/10.1016/j.arabjc.2019.08.001
Pasha, C. (2020). Determination of paracetamol in pharmaceutical samples by spectrophotometric method. Eclética Química, 45(3), 37-46. DOI:10.26850/1678-4618eqj.v45.3.2020.p37-46
Rahimpour, E., & Jouyban, A. (2023). Prediction of Paracetamol Solubility in Binary Solvents Using Reichardt’s Polarity Parameter Combined Model. Liquids, 3(4), 512-521. DOI: https://doi.org/10.3390/liquids3040032
Rahman, M. A. A., Elghobashy, M. R., Zaazaa, H. E., & El-Mosallamy, S. S. (2023). Novel analytical method based on chemometric models applied to UV–Vis spectrophotometric data for simultaneous determination of Etoricoxib and Paracetamol in presence of Paracetamol impurities. BMC chemistry, 17(1), 176. DOI:10.1186/s13065-023-01095-x
Salem, W. M. (2019). Sensitive determination of Paracetamol using Ferrocene Nanoparticles by Chitosan-Functionalized-Modified Carbon Past Electrode. Egyptian Journal of Chemistry, 62(4), 679-690. DOI: 10.21608/ejchem.2018.5231.1462
Selimoğlu, F., & Pinarcik, N. (2023). Spectrophotometric quantification of paracetamol and tramadol hydrochloride by chemometric calibration methods. Turkish Journal of Chemistry, 47(3), 633-645. DOI: 10.55730/1300-0527.3566
Thanoon, E. S. (2019). Spectrophotometric determination of paracetamol using diazotization coupling reaction. Rafidain Journal of Science, 28(2), 76-83. DOI:10.33899/rjs.2019.159979
Youssef, S. H., Mohamed, D., Hegazy, M. A. M., & Badawey, A. (2019). Analytical methods for the determination of paracetamol, pseudoephedrine and brompheniramine in Comtrex tablets. BMC chemistry, 13(1), 78. DOI: 10.1186/s13065-019-0595-6
Authors
Copyright (c) 2026 Vian S. Ismail, Sawza Abdulsalam Mohammed, Sumaya Tahir Juju, Khawla Muhammad Abdullah, Darya Husen Ibrahim, Rebin Khdir Mala, Hayam Fakher Fathulla, and Sumaya Mukhlis Sharif.

This work is licensed under a Creative Commons Attribution-NonCommercial-ShareAlike 4.0 International License.
Authors who publish with this journal agree to the following terms:
- Authors retain copyright and grant the journal right of first publication with the work simultaneously licensed under a Creative Commons Attribution License [CC BY-NC-SA 4.0] that allows others to share the work with an acknowledgment of the work's authorship and initial publication in this journal.
- Authors are able to enter into separate, additional contractual arrangements for the non-exclusive distribution of the journal's published version of the work, with an acknowledgment of its initial publication in this journal.
- Authors are permitted and encouraged to post their work online.